That is a reason not to teach about circular orbits and L = nħ. But second, it doesn’t conform to the essence of what Bohr actually did. Entitled On the Constitution of Atoms and Molecules, the article brought together for. In 1913, Niels Bohr published a groundbreaking paper that introduced a new way of understanding atomic phenomena ( 1 ). This, in itself, is not a reason to scrap it, for the historical development of quantum physics is certainly of interest. One hundred years ago, Niels Bohrs pioneering paper on the electronic structure of the hydrogen atom revolutionized atomic theory. Bohr atomic model and the models after that explain the properties of atomic electrons on the basis of certain allowed possible values. First it doesn’t conform to our present understanding of the hydrogen atom. This picture of the hydrogen atom is wrong in two senses. Niel Bohr’s Atomic Theory states that an atom is like a planetary model where electrons were situated in discretely energized orbits. (c) In the cathode ray, the beam (shown in yellow) comes from the cathode and is accelerated past the anode toward a fluorescent scale at the end of the tube. (b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. h E ( 1 n l o w 2 1 n h i g h 2) 13.6 eV. Thomson produced a visible beam in a cathode ray tube. Bohr explained the hydrogen spectrum in terms of electrons absorbing and emitting photons to change energy levels, where the photon energy is. Some of those models included Niels Bohr’s revised atomic model, Erwin Schrödinger’s equation, Max Planck’s constant h, and Rutherford’s experiments. Bohrs model calculated the following energies for an electron in the shell, n. 1913 was a year of highs and lows in physical sciences. 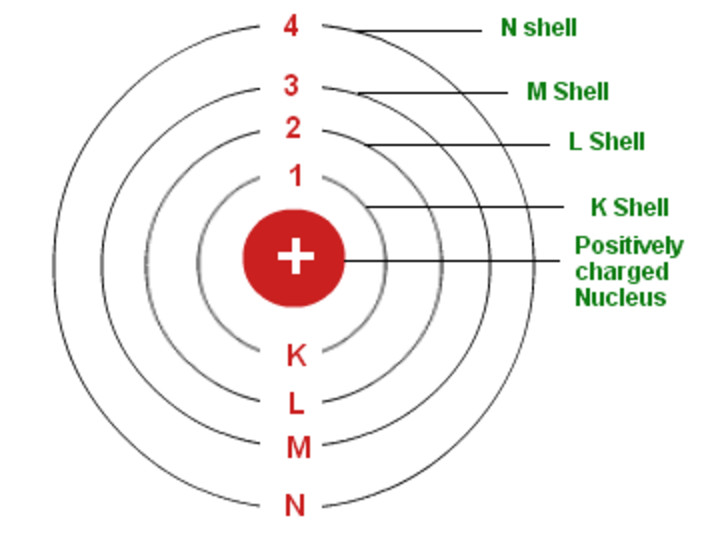
This “pedagogic” version postulates that an electron in a stationary state moves in a circular orbit with an angular momentum that is an integral multiple of h/2π ( L = nh/2π = nħ)- ħ for the lowest-energy state, 2 ħ for the next state, and so on. Bohr’s atomic theory is still widely used today, but many different models were tried that failed to make quantum mechanics work to be a better explanation of atoms. The events that led to the atomic theory unfolded as a series of scientific breakthroughs and puzzles. Unfortunately, the version of the Bohr atom that appears in many textbooks and is no doubt often presented to students is more wrong than right and may leave both teachers and students wondering why, more than a hundred years later, it is still being taught. This is an excellent idea, given the historic importance of Bohr’s 1913 work, which provided the bridge from Planck’s quantized interaction of matter and radiation (1900) to the full theory of quantum mechanics (1925-28). We have carefully curated multiple quizzes with varying difficulty levels for a well-rounded practice session. We have quizzes covering each and every topic of Physical Chemistry and other concepts of Chemistry. This atomic model is the modification of Rutherford’s atomic model (the nucleus is positively charged and is surrounded by electrons (negatively charged particles). Solve these Bohrs Model of an atom questions and sharpen your practice problem-solving skills. Many teachers like to introduce the Bohr atom toward the end of an introductory physics course.  
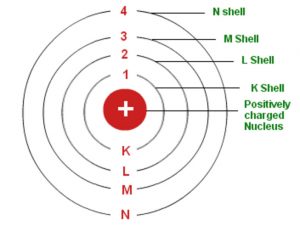
All substances, according to Dalton’s atomic theory, are made up of atoms, which are indivisible and. It stated that all matter was made up of small, indivisible particles known as ‘atoms’. A hydrogen atom with an electron in an orbit with n > 1 is therefore in an excited state, defined as any arrangement of electrons that is higher in energy than the ground state.Many teachers like to introduce the Bohr atom toward the end of an introductory physics course. In 1913, Danish physicist Neil Bohr proposed the Bohr atomic model based on Planck’s quantum theory of radiation. Dalton’s atomic theory was a scientific theory on the nature of matter put forward by the English physicist and chemist John Dalton in the year 1808.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
